Hazardous Pharmaceutical Waste Management Training | EPA 40 CFR Part 266 Subpart P
Pharmaceutical Waste Management Training Programs
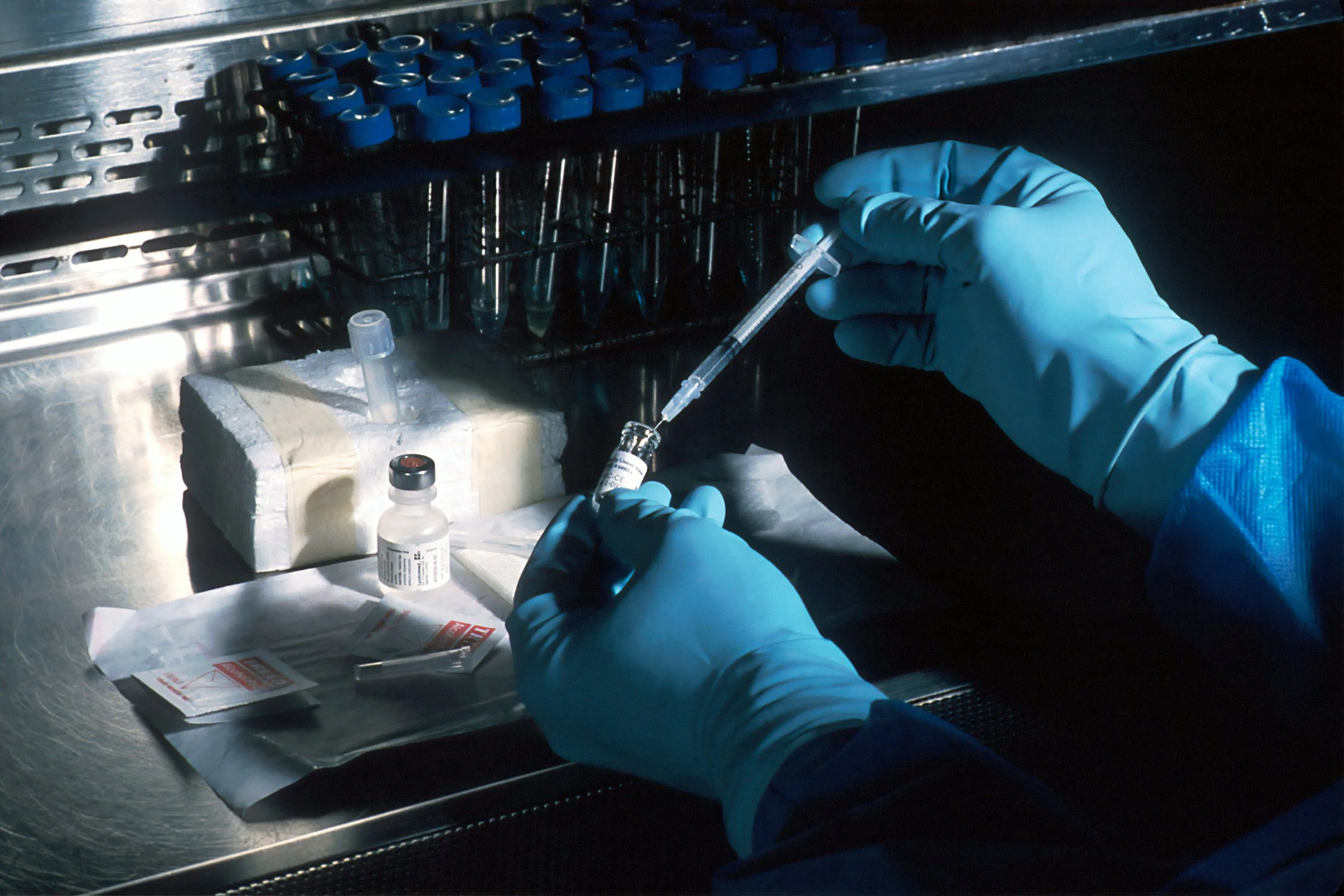
Pharmaceutical waste management is one of the most regulated and frequently cited areas in healthcare and research facilities. EDJ provides practical training that helps staff correctly identify, segregate, and manage pharmaceutical waste in accordance with federal, state, and facility-specific requirements.
Training focuses on applying regulations in daily workflows so pharmaceutical waste is handled correctly at the point of generation and remains compliant through disposal.
Pharmaceutical Waste Management Training Requirements
EPA RCRA regulations (40 CFR Parts 261 and 266 Subpart P), state-specific pharmaceutical waste rules, DEA requirements where applicable, and healthcare accreditation standards.
Pharmaceutical waste training is essential for facilities that generate expired, unused, or partially used medications. Improper handling can lead to regulatory violations, environmental contamination, and safety risks to staff and patients.
Topics Covered in Pharmaceutical Waste Training
Pharmaceutical Waste Identification
Hazardous versus non-hazardous pharmaceuticals
P-listed, U-listed, and characteristic wastes
Acute hazardous pharmaceutical identification
State-specific classifications
Identifying hazardous drugs (NIOSH)
Waste Segregation and Handling
Point-of-generation segregation
Container compatibility and labeling
Storage time limits and accumulation practices
Spill prevention and response
Regulatory Compliance
EPA Subpart P requirements
RCRA generator responsibilities
Documentation and recordkeeping
Coordination with disposal vendors
Workplace Safety
Exposure risks and prevention
PPE requirements
Emergency response considerations
Training documentation and competency tracking
Department Specific Applications
Especially relevant for healthcare and research environments:
Pharmacy operations
Nursing units and patient care areas
Surgical and procedural areas
Laboratories and research facilities
Environmental services workflows
In partnership with Global Hazmat, Inc.
Who Is Required to Complete Pharmaceutical Waste Management Training?
Employees Involved in Pharmaceutical Waste Handling
Pharmacy staff
Nursing and clinical personnel
Environmental services teams
Laboratory and research staff
Waste handling and compliance personnel
What Employees Must Be Able to Demonstrate
Identify pharmaceutical waste correctly
Segregate waste at the point of generation
Apply facility-specific procedures with confidence
Reduce regulatory risk during inspections
Training is tailored to actual processes used within each department to ensure consistency across the facility.
Pharmaceutical waste compliance depends on correct decisions made at the point of care, not after the fact.
Need More Services?
If your needs extend beyond training into system development, program evaluation, or regulatory alignment, our Compliance Solutions provide the structure and oversight to support long-term compliance.